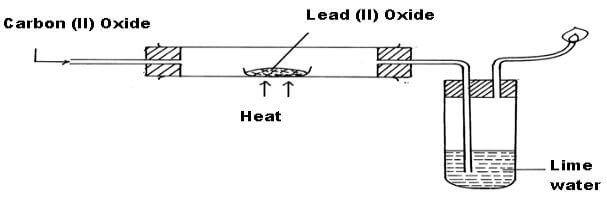
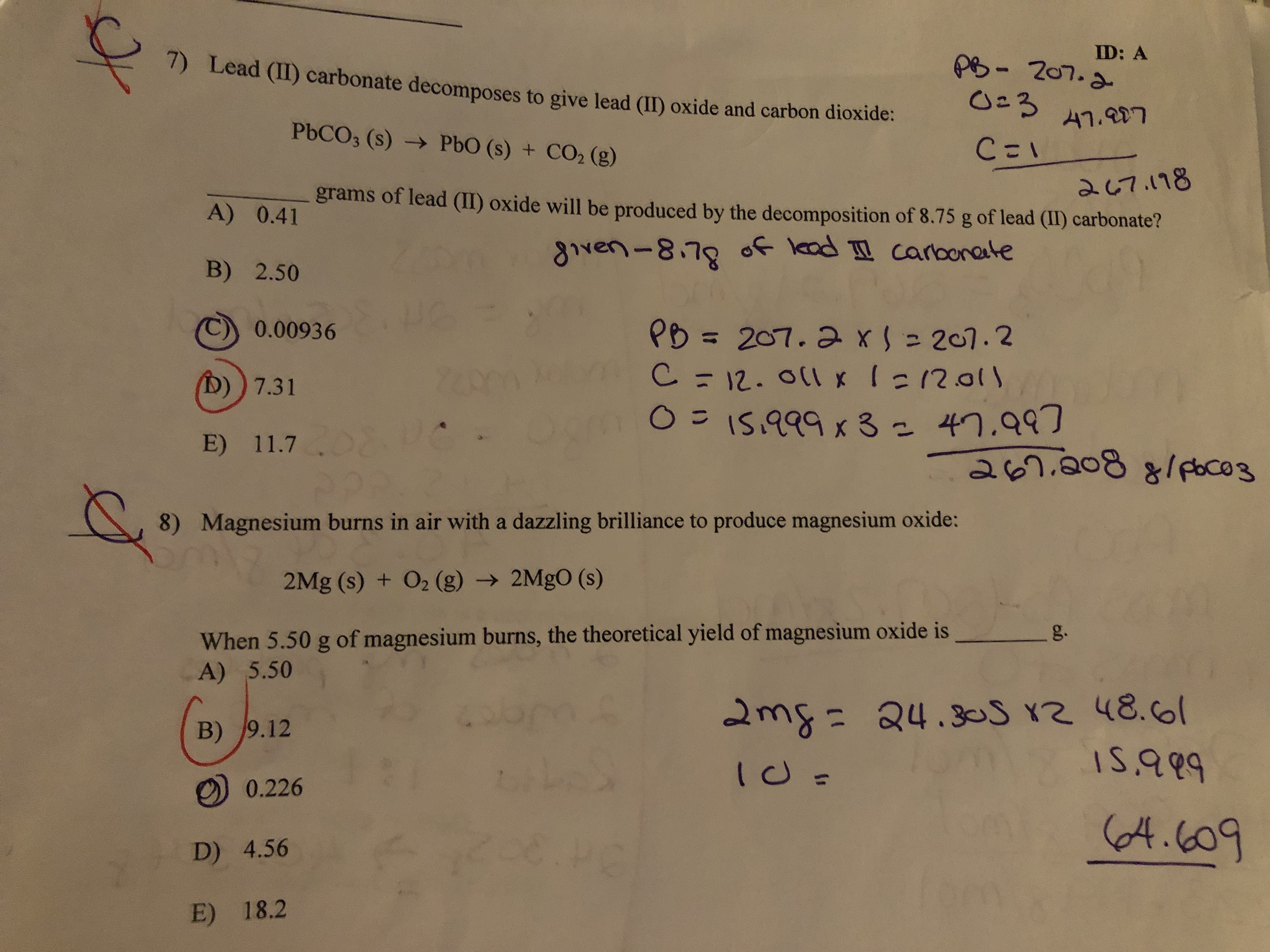SOLVED: 51R) Lead (Il) carbonate decomposes to give lead (II) oxide and carbon dioxide; PbCOz (s) PbO (s) CO2 (5) How many grams of lead (Il) oxide will be produced by the
Quiz (Extraction of Metals) 1. Metals are usually extracted from their ores before use. State the method of extraction for the f
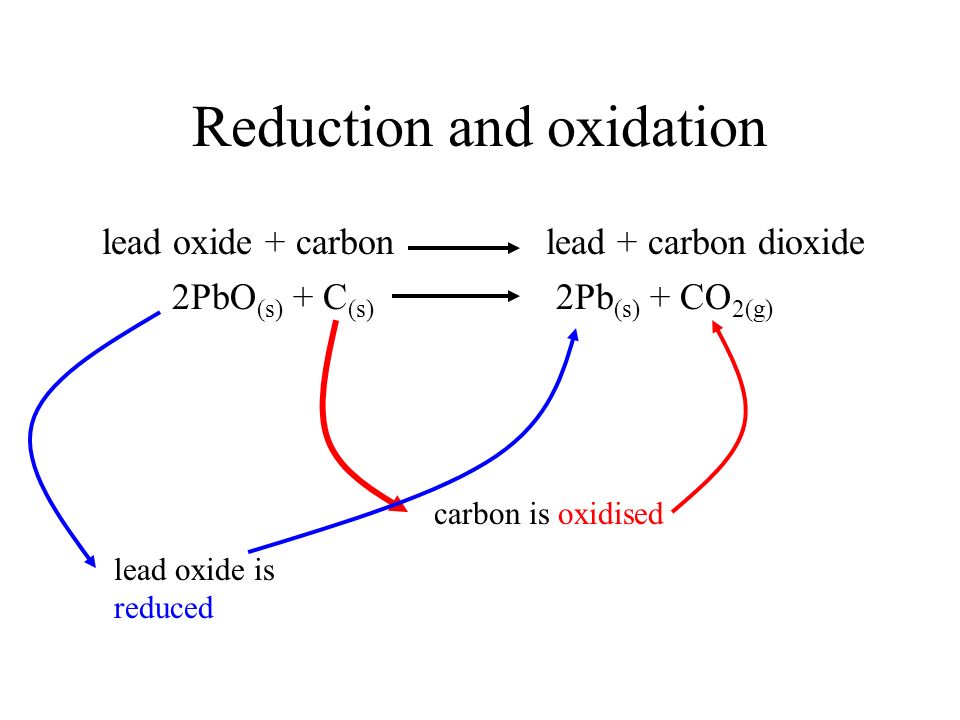
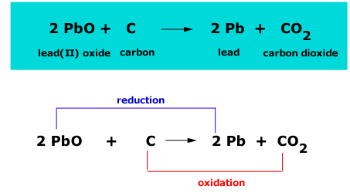
![For the given reaction,\\[2PbO + C \\to Pb + C{O_2}\\] name the oxidized substance, reduced substance, reducing agent, and oxidizing agent in this reaction. For the given reaction,\\[2PbO + C \\to Pb + C{O_2}\\] name the oxidized substance, reduced substance, reducing agent, and oxidizing agent in this reaction.](https://www.vedantu.com/question-sets/407a8186-d96f-4ae5-9b1b-a35a89f410b32918307568284777948.png)
For the given reaction,\\[2PbO + C \\to Pb + C{O_2}\\] name the oxidized substance, reduced substance, reducing agent, and oxidizing agent in this reaction.
18 Which of the statements about the reaction below are incorrect? 2PbO(s)+C(s)→ 2Pb(s)+CO2(g) (a) Lead is getting reduced (b) Carbon Dioxide is getting oxidised (c) Carbon is getting oxidised (d) Lead oxide
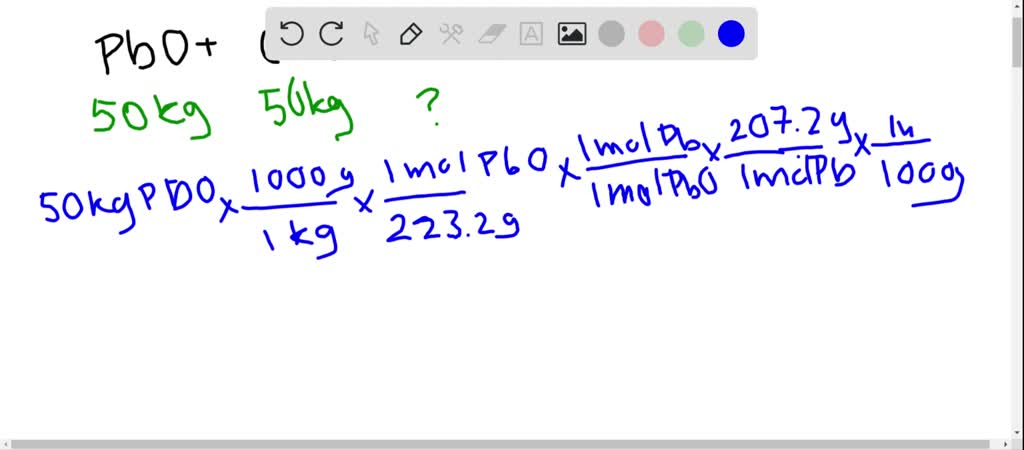
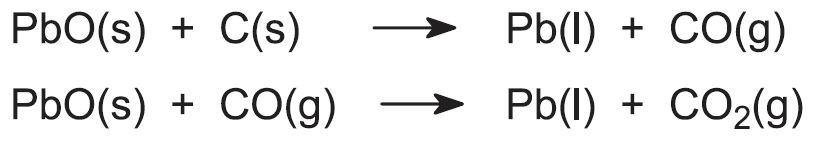
SOLVED:Lead(II) oxide from an ore can be reduced to elemental lead by heating in a furnace with carbon. PbO(s)+C(s) →Pb(l)+CO(g) Calculate the expected yield of lead if 50.0 kg of lead oxide

Preparation of High Purity Lead Oxide from Spent Lead Acid Batteries via Desulfurization and Recrystallization in Sodium Hydroxide | Industrial & Engineering Chemistry Research

Which of the statements about the reaction below are incorrect ? 2PbO(s) + C(s) → 2Pb(s) + CO2(g) (a) Lead is getting reduced.(b) Carbon dioxide is getting oxidised.(c) Carbon is getting oxidised.(d)
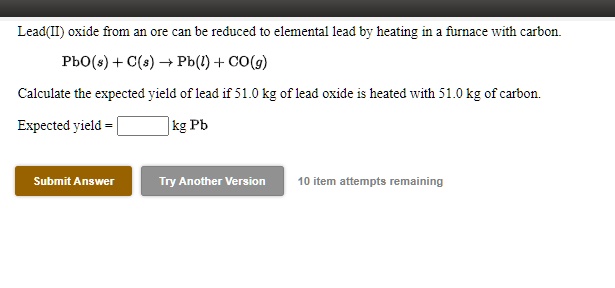
SOLVED: Lead(Il) oxide from an ore can be reduced to elementa) lead by heating in fnace With carbon Pbo(s) + C(s) = Pb(l) + CO(g) Calculate the expected yield of lead if